We depart at the crack of dawn as we do every Monday. Our destination is the animal laboratory housed in the building of the medical and research centre where we go to look in on the laboratory mice. The animals in our segment of cages look like ordinary mice. Some are still sleeping through the morning hours, others eating their breakfast. But these are no ordinary mice – their unique qualities are merely hidden from the naked eye. They are hybrid mice in which scientists have developed a human immune system, meaning that the majority of cells in the immune systems of our mice are of human origin. Moreover, each of our hybrid mice has had a human tumour implanted inside of it. The tumours are still in the early phase of development, their impact not yet felt by the animals. These mice are part of a study exploring immunotherapy of cancer, a field which has recently been experiencing a renaissance. This type of therapy which triggers the patient's immune system has proven to be a very effective method of combating various kinds of cancer.
The Immune Response in Cancer
The successful understanding of the processes that the immune system undergoes in response to cancer is a crucial step in the development of effective immunotherapies. When normal cells transform into tumour cells, they develop the ability to influence the immune system and thereby avoid its surveillance functions. Both cytotoxic T cells and natural killer (NK) cells play an important role in the destruction of tumour cells. Cytotoxicity is the process by which activated lymphocytes destroy a tumour cell at the instant of contact. In the early phases of cancer, the immune system is still capable of recognizing and destroying tumour cells, but, as the cancer develops, the immune system loses the ability to control its growth. At some point, tumour cells begin to rearrange the environment, diminishing the activities of cytotoxic immune cells and their ability both to proliferate and to recognize tumour antigens. Tumour antigens are specific proteins on the surface of tumour cells. Cytotoxic T cells are capable of recognizing such proteins, which is what allows them to efficiently destroy tumour cells. However, in addition to tumour antigens, the surface of a tumour cell also contains proteins that inhibit the functioning of cytotoxic lymphocytes. These proteins, called immune checkpoint inhibitors, play an important role in the prevention of autoimmunity, and in the case of tumours cells, can block their destruction. The goal of immunotherapy is to activate cytotoxic cells and thus support the activities of the immune system. This is done by suppressing immune checkpoint inhibitors, generating signalling molecules associated with inflammation, and stimulating the immune system's recognition of the proteins on the surface of tumour cells.
The Origin of Tumours – Tumour Stem Cells
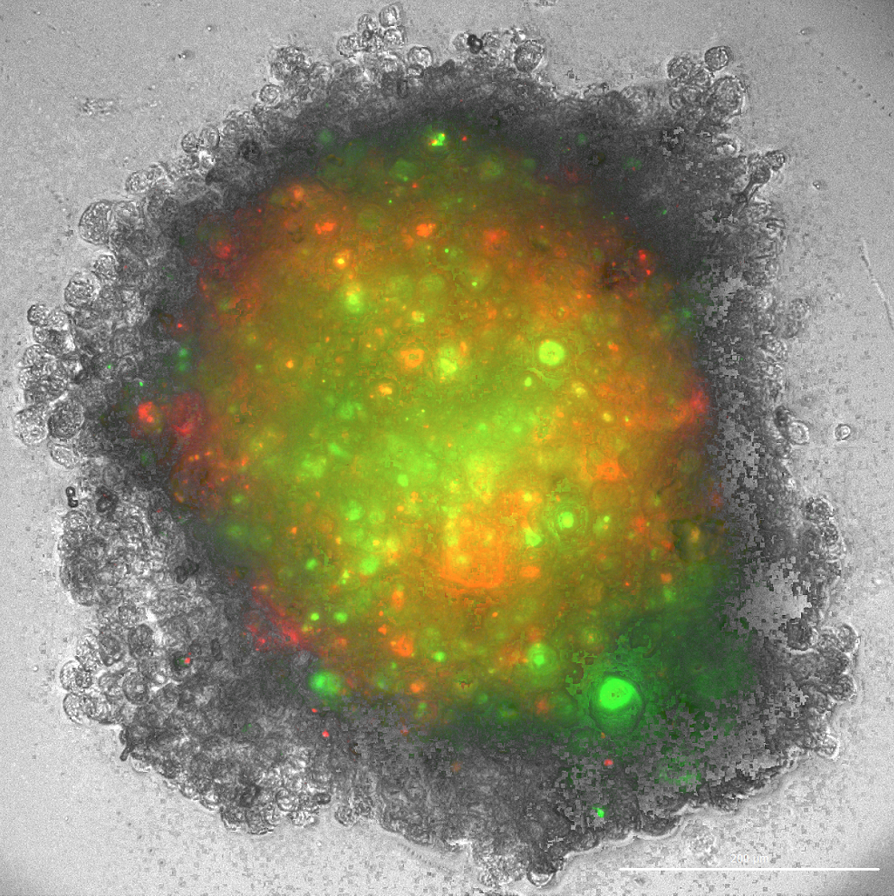
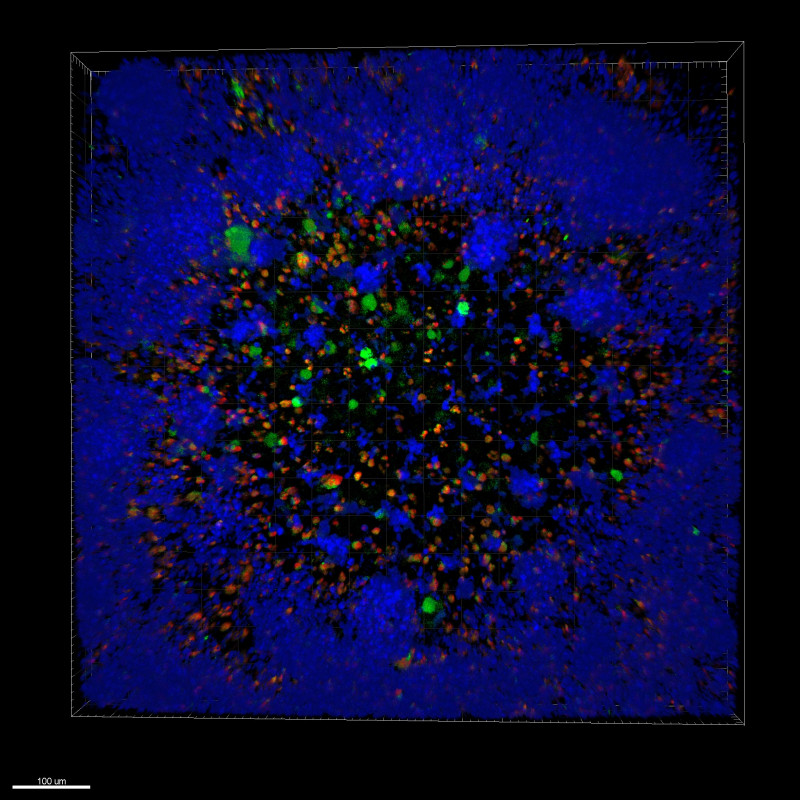
After the treatment of cancer patients with radiation therapy or chemotherapy, a number of tumour cells remain in the afflicted tissue that are resistant to treatment. Because the tumour's mass can no longer be detected, the cancer appears to be entirely eliminated, but, in fact, the surviving tumour cells may cause a regrowth of the tumour or even metastasize with the passage of time. Scientists have isolated the surviving cells in tumorous tissue and concluded that these populations of cells contain activated genes and signalling pathways that are characteristic of normal stem cells. They also discovered that these tumour cells contain the ability to self-renew and differentiate, another quality associated with stem cells. These findings suggest that it is the stem cells of tumours that are responsible for keeping the tumour itself alive, and moreover it is their capacities for self-renewal and differentiation that may cause the emergence of a new tumour with the same features as the original tumour. If we isolate tumour stem cells from a patient and implant them in laboratory mice, the mice develop cancerous forms that are very similar to the patient’s initial tumour. These stem cells are resistant to chemotherapy as they contain effective mechanisms for repairing damage to their DNA. This enables them to survive in the wake of chemotherapy and thus to preserve the tumour. However, these properties also make them an ideal target for the suppression of tumour growth. Of course, the removal of tumour stem cells is far from a simple procedure. Tumour stem cells are extremely heterogeneous and exhibit a range of different proteins on their surface. Moreover, they are highly adaptable to signals from their environment, rapidly transforming their characteristics and developing resistance to treatments which makes it extremely difficult to target them within the tumour.
Is Immunotherapy with Natural Killer (NK) Cells the Solution?
Tumour stem cells are highly effective at evading responses from the immune system. For example, the molecules of the major histocompatibility complex (MCH) I, that makes it possible for cytotoxic T cells to recognize and destroy foreign bodies, are not expressed on the surface of tumour stem cells. NK cells are a part of the innate immune response and are the only cells within the immune system capable of recognizing and destroying tumour stem cells without the prior antigen presentation. Upon contact with a tumour stem cell, NK cells recognize the absence of molecules of the major histocompatibility complex (MCH) I. This causes them to activate and releasing cytotoxic granules that destroy the tumour cell. In addition, upon activation, NK cells release the signalling molecule interferon gamma (IFN-γ) into their environment, which then activates other cells of the immune system triggering an immune response against the tumour. Research on hybrid mice implanted with human tumours of the pancreas and the oral cavity shows that the injection of NK cells from healthy human donors significantly reduces the growth of the tumours in the mice, and activated NK cells demonstrate a remarkable ability to destroy the stem cells of the tumours.
Researchers at Slovenia's National Institute of Biology (NIB) have spent several years studying brain tumours. Brain tumours of the astrocytic origin – also known as glioblastoma multiforme – are the most malignant of all brain tumours, and there is no effective treatment for them. Immunotherapy with NK cells provides a potential approach to the treatment of glioblastomas, particularly given the fact that the resurgence of glioblastomas is in most cases caused by the tumour stem cells. Currently, we are collaborating with the University of California Los Angeles in order to develop a range of pre-clinical cellular and animal models that would study the efficiency of NK cells in destroying the stem cells of tumours. Our research indicates that NK cells may be highly effective at destroying fast-growing stem cells of brain tumours. NK cells demonstrate the additional benefit of releasing large quantities of the molecule interferon gamma that triggers an additional response from the immune system.
To return to the mice in our introduction, research on tumour cell cultures and hybrid mice with human tumours and human immune systems has provided enormous benefits in pre-clinical studies of immunotherapy as these models replicate the interaction between the immune system and a tumour within the human body. In developing cancer treatments, we are seeking to identify approaches that target extremely malignant and therapy-resistant tumour stem cells. In this regard, immunotherapy with NK cells presents an excellent avenue for further research.
Translated by Erica J. Debeljak